Put EXOGEN to work for your patients.
Behind every fracture is the story of a patient hoping to heal quickly and resume an active life.
Here’s one of those stories.
Case Study Background
Patient
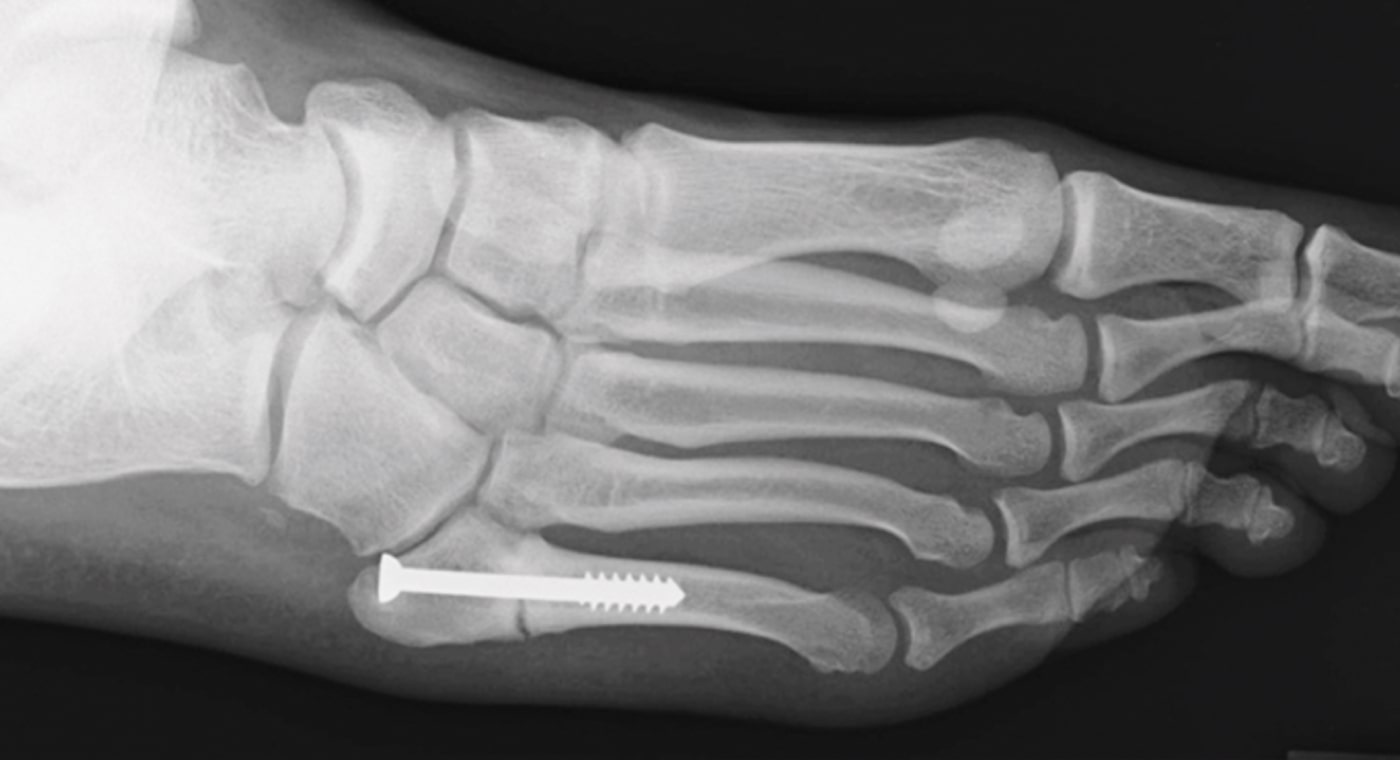
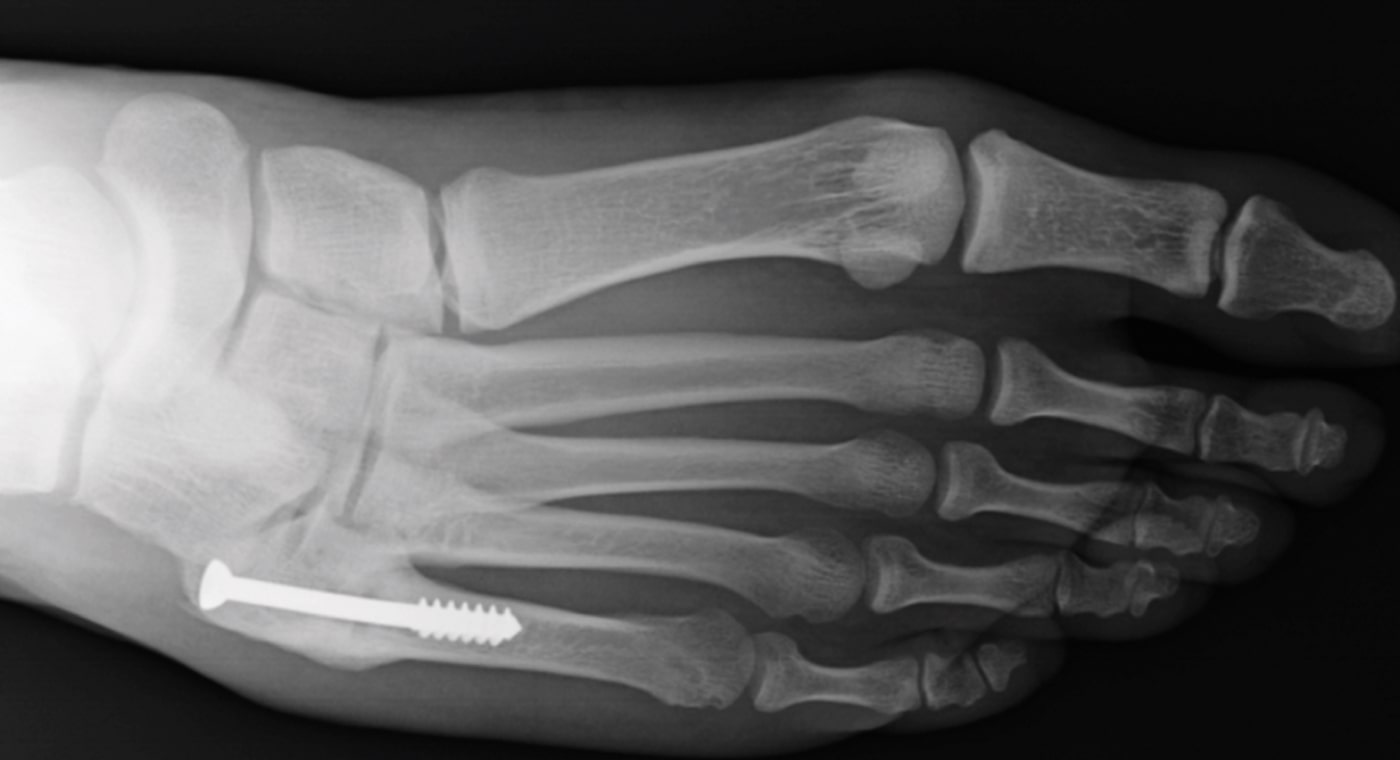
20-year-old male college football player with a history of Jones fracture, two prior surgeries, and no comorbidities.
Initial History
The patient was involved in a motor vehicle accident in which he refractured the fifth metatarsal on his right foot.
The patient was treated conservatively with a cast and boot.

8 Months Post-Op
- The patient returned to playing football.
Source: Robert Anderson, MD, OrthoCarolina Foot & Ankle Institute Charlotte, NC
Individual results may vary. Patients should consult with a qualified physician to determine if this product is right for them.
Put EXOGEN to work for your
fracture patients.
Your patients can do more than just wait to heal. Only 20 minutes a day of safe, painless treatment with EXOGEN can significantly accelerate the body’s natural bone-healing process, and help repair fractures that have failed to heal and might otherwise require surgery.1-3,68
EXOGEN has been shown to help the fracture healing process for patients with certain comorbidities that can delay fracture healing, putting the fractures at risk of nonunion.35
Let Us Show You What EXOGEN Can Do
Summary of Indications for Use:
The EXOGEN Ultrasound Bone Healing System is indicated for the non-invasive treatment of established nonunions* excluding skull and vertebra. The EXOGEN device has also been reported as effective as an adjunctive non-invasive treatment of established nonunions in patients:
- With internal or external fracture fixation hardware present. EXOGEN cannot penetrate metal and therefore should not be applied directly over hardware.
- Undergoing treatment for infection at the fracture site. EXOGEN is not intended to treat the infection.
- Believed to have diminished bone quality. EXOGEN is not intended to treat diminished bone quality.
In addition, EXOGEN is indicated for accelerating the time to a healed fracture for fresh, closed, posteriorly displaced distal radius fractures and fresh, closed or Grade I open tibial diaphysis fractures in skeletally mature individuals when these fractures are orthopedically managed by closed reduction and cast immobilization.
There are no known contraindications for the EXOGEN device. Safety and effectiveness have not been established for individuals lacking skeletal maturity, pregnant or nursing women, patients with cardiac pacemakers, on fractures due to bone cancer, or on patients with poor blood circulation or clotting problems. Some patients may be sensitive to the ultrasound gel.
*A nonunion is considered to be established when the fracture site shows no visibly progressive signs of healing.
Full prescribing information can be found in product labeling, at EXOGEN.com, or by calling Customer Service at 1-800-836-4080.
All revisions of the Instructions for Use can be made available upon request. Contact [email protected] or call 1-800-836-4080 to request an electronic copy.