What to expect about EXOGEN’s coverage and benefits
When choosing the best options to treat a fracture, you probably consider efficacy first and foremost. However, other factors also may be important, such as the cost of surgical intervention, as well as the patient’s out-of-pocket cost, insurance coverage, and work situation.
These facts about EXOGEN can help you make the best choice for your fracture patients.
Patient costs for EXOGEN
Patients may be concerned about their out-of-pocket costs for EXOGEN. The Fracture Care team is available at 800-836-4080 to answer questions about cost and coverage.
Note: EXOGEN is covered by many major health insurance plans, such as:13-16,79
• Aetna® • Cigna® • Humana • UnitedHealthcare® • Blue Cross® Blue Shield®
In addition, the EXOGEN Rewards program offers reimbursement of up to $500 of a patient’s out-of-pocket costs for the EXOGEN device. (Reimbursement cannot exceed the patient’s actual out-of-pocket costs.)
Surgery vs EXOGEN
Research has established that EXOGEN’s low-intensity pulsed ultrasound (LIPUS) heals chronic nonunions without the need for revision surgery.29,35
For example, Zura et al, reported heal rates of patients (N = 767) who had established nonunions treated with LIPUS:35
86.2%
(N = 767) overall heal rate
82.7%
(N = 98) heal rate among patients with a chronic nonunion at least 5 years old
63.2%
(N = 19) heal rate among patients with a chronic nonunion more than 10 years old
HCPCS Code: E0760
Description: EXOGEN Ultrasound Bone Healing System by Bioventus
Let’s Show You What EXOGEN Can Do
Individual results may vary. Patients should consult with a qualified physician to determine if this product is right for them.
Summary of Indications for Use:
The EXOGEN Ultrasound Bone Healing System is indicated for the non-invasive treatment of established nonunions* excluding skull and vertebra. The EXOGEN device has also been reported as effective as an adjunctive non-invasive treatment of established nonunions in patients:
- With internal or external fracture fixation hardware present. EXOGEN cannot penetrate metal and therefore should not be applied directly over hardware.
- Undergoing treatment for infection at the fracture site. EXOGEN is not intended to treat the infection.
- Believed to have diminished bone quality. EXOGEN is not intended to treat diminished bone quality.
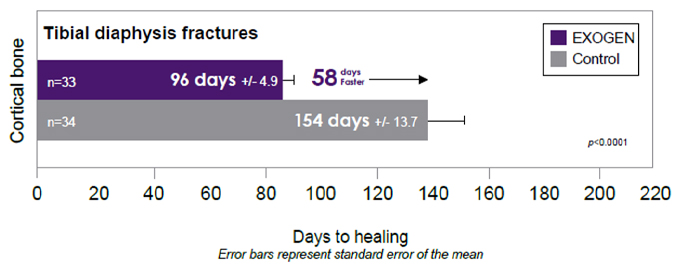
In addition, EXOGEN is indicated for accelerating the time to a healed fracture for fresh, closed, posteriorly displaced distal radius fractures and fresh, closed or Grade I open tibial diaphysis fractures in skeletally mature individuals when these fractures are orthopedically managed by closed reduction and cast immobilization.
There are no known contraindications for the EXOGEN device. Safety and effectiveness have not been established for individuals lacking skeletal maturity, pregnant or nursing women, patients with cardiac pacemakers, on fractures due to bone cancer, or on patients with poor blood circulation or clotting problems. Some patients may be sensitive to the ultrasound gel.
*A nonunion is considered to be established when the fracture site shows no visibly progressive signs of healing.
Full prescribing information can be found in product labeling, at EXOGEN.com, or by calling Customer Service at 1-800-836-4080.
All revisions of the Instructions for Use can be made available upon request. Contact [email protected] or call 1-800-836-4080 to request an electronic copy.