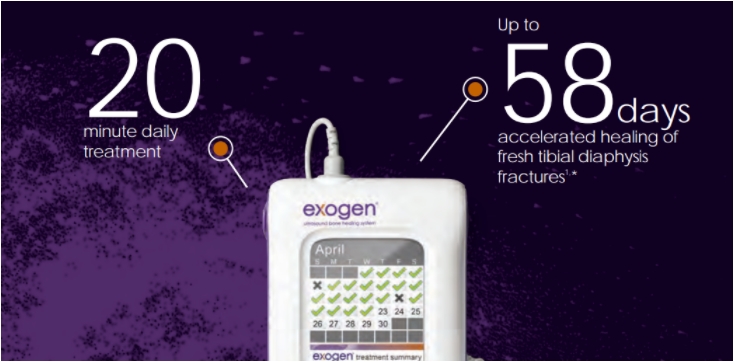
Design Evolution of EXOGEN Enhances Patient Compliance
N. Pounder PhD, Director Project Management - Bioventus LLC
EXOGEN Accelerates Healing of Fractures in Smokers
Dr Zura, Orthopaedic Surgeon LSU – LSU Health Sciences Center New Orleans
EXOGEN and Fracture Healing in Uncertain Times
Dr Zura, Orthopaedic Surgeon LSU – LSU Health Sciences Center New Orleans
EXOGEN Casting Application Techniques
Instruction for building a transducer port into a new cast or creating a window in an existing cast
EXOGEN Mechanism of Action Brochure
Scientific explanation of EXOGEN’s unique mechanism of action
Acute Fractures
- Acceleration of tibia and distal radius fracture healing in patients who smoke.
- Acceleration of tibial fracture-healing by non-invasive, low-intensity pulsed ultrasound.
- Accelerated healing of distal radial fractures with the use of specific, low-intensity ultrasound. A multicenter, prospective, randomized, double-blind, placebo-controlled study.
Nonunion Fractures
- Indications and results for the Exogen™ ultrasound system in the management of nonunion: a 59-case pilot study.
- Low-intensity pulsed ultrasound in the treatment of nonunions.
- Low-intensity pulsed ultrasound: effects on nonunions.
- Improved healing response in delayed unions of the tibia with low-intensity pulsed ultrasound: results of a randomized sham-controlled trial.
- The evaluation of the healing rate of subtalar arthrodeses, part 2: the effect of low-intensity ultrasound stimulation.
- Compound high-energy limb fractures with delayed union: our experience with adjuvant ultrasound stimulation (exogen).
- Failure of surgery for scaphoid non-union is associated with smoking.
- Treatment of chronic (>1 year) fracture nonunion: heal rate in a cohort of 767 patients treated with low-intensity pulsed ultrasound (LIPUS).
Other
- Mode & mechanism of low intensity pulsed ultrasound (LIPUS) in fracture repair.
- Prolonged endochondral bone healing in senescence is shortened by low-intensity pulsed ultrasound in a manner dependent on COX-2.
- Sources of information influencing decision-making in orthopaedic surgery – an international online survey of 1147 orthopaedic surgeons.
- Design evolution enhances patient compliance for low-intensity pulsed ultrasound device usage.
REWARDING YOUR PATIENTS TO DRIVE COMPLIANCE AND BETTER OUTCOMES
EXOGEN Rewards is a reimbursement program that encourages patients to comply with their treatment regimen and achieve successful bone healing.
The program reimburses eligible patients up to $500 of their out-of-pocket costs for the EXOGEN device. Reimbursement cannot exceed a patient’s actual out-of-pocket costs.
ELIGIBILITY
Not all patients are eligible for EXOGEN Rewards. Terms and conditions apply.
To be eligible for EXOGEN Rewards, the patient must be 95% compliant with daily treatment, and the patient must either:
-
- Use EXOGEN daily for a minimum of 120 days* (recorded automatically by the device);
Or
-
- Provide proof of healing, if healing occurs prior to 120 days (the prescribing healthcare provider must complete the Bioventus Attestation Form.
Other eligibility requirements apply, as well. For example:
-
- The patient must call Customer Service within 30 days after their last treatment.
EVALUATION
The EXOGEN device contains an internal usage monitor that records the date, time, and duration of each treatment session. Bioventus will use the monitor to confirm whether the patient complied with the treatment regimen at least 95% of the time.
EXCLUSIONS
Not all users are eligible for EXOGEN Rewards. Program benefits will not be available for:
-
- An EXOGEN device that has been modified or altered
- An EXOGEN device that was not sent to the patient directly from Bioventus
- A patient who was not prescribed EXOGEN
- Patients with a denied EXOGEN order
- Costs other than the patient’s out-of-pocket payment to Bioventus
- EXOGEN treatment outside the United States
- Patients with any federal, state, or other government plans, including, but not limited to, Medicare (including a Medicare Part D or Medicare Advantage plan), Medicaid, TRICARE/CHAMPUS, Veterans Administration or Department of Defense health coverage, the Puerto Rico Government Health Insurance Plan, or any other federal or state healthcare programs or pharmaceutical assistance program
SUPPORT
Bioventus Customer Service is available to answer questions and help patients stay consistent with their EXOGEN treatments.
Call 1-800-836-4080.
*Terms and conditions apply.
- The EXOGEN Post-Market Data App contains real-world bone healing statistics, which can be used to benchmark your specific patients.
- It’s our free smartphone app that automatically tells you the heal rate associated with common fractures, factoring in patient risk factors and other relevant clinical considerations.
- Let the EXOGEN Post-Market Data App help you find the next appropriate patient.
Summary of Indications for Use
The EXOGEN Ultrasound Bone Healing System is indicated for the non-invasive treatment of established nonunions* excluding skull and vertebra. The EXOGEN device has also been reported as effective as an adjunctive non-invasive treatment of established nonunions in patients:
- With internal or external fracture fixation hardware present. EXOGEN cannot penetrate metal and therefore should not be applied directly over hardware.
- Undergoing treatment for infection at the fracture site. EXOGEN is not intended to treat the infection.
- Believed to have diminished bone quality. EXOGEN is not intended to treat diminished bone quality.
In addition, EXOGEN is indicated for accelerating the time to a healed fracture for fresh, closed, posteriorly displaced distal radius fractures and fresh, closed or Grade I open tibial diaphysis fractures in skeletally mature individuals when these fractures are orthopedically managed by closed reduction and cast immobilization.
There are no known contraindications for the EXOGEN device. Safety and effectiveness have not been established for individuals lacking skeletal maturity, pregnant or nursing women, patients with cardiac pacemakers, on fractures due to bone cancer, or on patients with poor blood circulation or clotting problems. Some patients may be sensitive to the ultrasound gel.
*A nonunion is considered to be established when the fracture site shows no visibly progressive signs of healing.
Full prescribing information can be found in product labelling, at EXOGEN.com, or by calling Customer Service at 1-800-836-4080.